| GSTZ1 | |||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Identifiers | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Aliases | GSTZ1, GSTZ1-1, MAAI, MAI, glutathione S-transferase zeta 1, MAAID | ||||||||||||||||||||||||||||||||||||||||||||||||||
| External IDs | OMIM: 603758 MGI: 1341859 HomoloGene: 7747 GeneCards: GSTZ1 | ||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikidata | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
Glutathione S-transferase Zeta 1 (also known as maleylacetoacetate isomerase) is an enzyme that in humans is encoded by the GSTZ1 gene on chromosome 14.[5][6][7]
This gene is a member of the glutathione S-transferase (GSTs) super-family, which encodes multifunctional enzymes important in the detoxification of electrophilic molecules, including carcinogens, mutagens, and several therapeutic drugs, by conjugation with glutathione. This enzyme also plays a significant role in the catabolism of phenylalanine and tyrosine. Thus, defects in this enzyme may lead to severe metabolic disorders, including alkaptonuria, phenylketonuria and tyrosinaemia, and new discoveries may allow the enzyme to protect against certain diseases related to oxidative stress.[7]
Structure
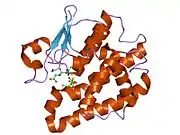
Glutathione S-transferase Zeta 1 (GSTZ1) has a predominantly hydrophobic dimer, just like many other GST members. It is composed of 24.2 kDa subunits and it consists of an N-terminal thioredoxin-like domain and a C-terminal all alpha-helical domain. Both of these domains are intertwined by a linker region between amino acids 85 and 91. The active site of this enzyme is much smaller and more polar than that of other family members of GST, which allows for GSTZ1 to be more selective in terms of substrates. Also, the C-terminus is truncated and the GSTZ1 enzyme lacks the normal V-shaped dimer interface which are usually common in other GSTs.[8] As for the GSTZ1 gene, it is located on chromosome 14q24.3, has 12 exons, and is approximately 10 kb long.[7] GSTZ1 also contains a distinct motif (Ser14–Ser15–Cys16) which is seen as the active center in catalysis.[9]
Function
GSTZ1 is predominantly found in liver cells; more specifically, it is localized in both the cytosol and the mitochondria.[10] GSTZ1 is essentially known for catalyzing glutathione-dependent isomerization of maleylacetoacetate to fumarylacetoacetate, which is the second-to-last step in the vital phenylalanine and tyrosine degradation pathway. It is the only enzyme in the GST family that catalyses a significant process in intermediary metabolism and it ensures that this enzyme can be found in a variety of species from humans to bacteria.[11] Another function of the GSTZ1 is that it is in control of the biotransformation of alpha-haloacids, like dichloroacetic acid (DCA), to glyoxylic acid. This prevents the buildup of DCA, which can lead to asymptomatic hepatotoxicity and a reversible peripheral neuropathy.[10] Both functions for this enzyme requires the presence of glutathione (GSH) in order to work.[9]
Clinical Significance
Deficiencies in any of the enzymes in the catabolism of phenylalanine and tyrosine, like GSTZ1, has led to diseases such as alkaptonuria, phenylketonuria, and several forms of tyrosinemia.[8] A lack of GSTZ1, specifically, leads to the amalgamation of maleylacetoacetate and succinylacetone which has been observed to cause oxidative stress. Also, scarcities have been seen to alter the metabolism of certain drugs and xenobiotics in mice.[12]
Most importantly, researchers have successfully genetically engineered GSTZ1 to mimic one of the most significant antioxidant enzymes, glutathione peroxidase (GPX). GPX is most known for its role to protect cells and tissues against oxidative damage by catalyzing the reduction of hydroperoxides using GSH as a reducing substrate and blocking the radical reaction caused by lipid peroxides. By protecting against this oxidative damage, GPX essentially prevents against degenerative diseases such as atherosclerosis, myocardial ischemia, heart failure, diabetes, pulmonary fibrosis, neurodegenerative disorders, and Alzheimer’s disease. However, because of GPX’s poor stability and paucity, it cannot be used in clinical studies and other methods must be considered. The newfound seleno-hGSTZ1–1 (or the engineered GSTZ1 enzyme) has a high GPX activity and a very similar reaction mechanism to that of GPX.[13]
Interactions
GSTZ1 has been seen to interact with:
- α-haloacids[9]
- GSH[10]
- Maleylacetoacetate[11]
References
- 1 2 3 GRCh38: Ensembl release 89: ENSG00000100577 - Ensembl, May 2017
- 1 2 3 GRCm38: Ensembl release 89: ENSMUSG00000021033 - Ensembl, May 2017
- ↑ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ↑ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ↑ Board PG, Baker RT, Chelvanayagam G, Jermiin LS (Dec 1997). "Zeta, a novel class of glutathione transferases in a range of species from plants to humans". The Biochemical Journal. 328. 328 (3): 929–35. doi:10.1042/bj3280929. PMC 1219006. PMID 9396740.
- ↑ Fernández-Cañón JM, Peñalva MA (Jan 1998). "Characterization of a fungal maleylacetoacetate isomerase gene and identification of its human homologue". The Journal of Biological Chemistry. 273 (1): 329–37. doi:10.1074/jbc.273.1.329. hdl:10261/169859. PMID 9417084.
- 1 2 3 "Entrez Gene: GSTZ1 glutathione transferase zeta 1 (maleylacetoacetate isomerase)".
- 1 2 Polekhina G, Board PG, Blackburn AC, Parker MW (Feb 2001). "Crystal structure of maleylacetoacetate isomerase/glutathione transferase zeta reveals the molecular basis for its remarkable catalytic promiscuity". Biochemistry. 40 (6): 1567–76. doi:10.1021/bi002249z. PMID 11327815.
- 1 2 3 Ricci G, Turella P, De Maria F, Antonini G, Nardocci L, Board PG, Parker MW, Carbonelli MG, Federici G, Caccuri AM (Aug 2004). "Binding and kinetic mechanisms of the Zeta class glutathione transferase". The Journal of Biological Chemistry. 279 (32): 33336–42. doi:10.1074/jbc.M404631200. PMID 15173170.
- 1 2 3 Li W, Gu Y, James MO, Hines RN, Simpson P, Langaee T, Stacpoole PW (Feb 2012). "Prenatal and postnatal expression of glutathione transferase ζ 1 in human liver and the roles of haplotype and subject age in determining activity with dichloroacetate". Drug Metabolism and Disposition. 40 (2): 232–9. doi:10.1124/dmd.111.041533. PMC 3263939. PMID 22028318.
- 1 2 Ketterer B (Oct 2001). "A bird's eye view of the glutathione transferase field". Chemico-Biological Interactions. 138 (1): 27–42. doi:10.1016/s0009-2797(01)00277-0. PMID 11640913.
- ↑ Blackburn AC, Matthaei KI, Lim C, Taylor MC, Cappello JY, Hayes JD, Anders MW, Board PG (Feb 2006). "Deficiency of glutathione transferase zeta causes oxidative stress and activation of antioxidant response pathways". Molecular Pharmacology. 69 (2): 650–7. doi:10.1124/mol.105.018911. PMID 16278372. S2CID 18371360.
- ↑ Yin L, Song J, Board PG, Yu Y, Han X, Wei J (Jan 2013). "Characterization of selenium-containing glutathione transferase zeta1-1 with high GPX activity prepared in eukaryotic cells". Journal of Molecular Recognition. 26 (1): 38–45. doi:10.1002/jmr.2241. PMID 23280616. S2CID 20923547.
Further reading
- Ketterer B (Oct 2001). "A bird's eye view of the glutathione transferase field". Chemico-Biological Interactions. 138 (1): 27–42. doi:10.1016/S0009-2797(01)00277-0. PMID 11640913.
- Tong Z, Board PG, Anders MW (Apr 1998). "Glutathione transferase zeta catalyses the oxygenation of the carcinogen dichloroacetic acid to glyoxylic acid". The Biochemical Journal. 331. 331 (2): 371–4. doi:10.1042/bj3310371. PMC 1219363. PMID 9531472.
- Tong Z, Board PG, Anders MW (Nov 1998). "Glutathione transferase zeta-catalyzed biotransformation of dichloroacetic acid and other alpha-haloacids". Chemical Research in Toxicology. 11 (11): 1332–8. doi:10.1021/tx980144f. PMID 9815194.
- Blackburn AC, Woollatt E, Sutherland GR, Board PG (1999). "Characterization and chromosome location of the gene GSTZ1 encoding the human Zeta class glutathione transferase and maleylacetoacetate isomerase". Cytogenetics and Cell Genetics. 83 (1–2): 109–14. doi:10.1159/000015145. PMID 9925947. S2CID 22835884.
- Fernández-Cañón JM, Hejna J, Reifsteck C, Olson S, Grompe M (Jun 1999). "Gene structure, chromosomal location, and expression pattern of maleylacetoacetate isomerase". Genomics. 58 (3): 263–9. doi:10.1006/geno.1999.5832. PMID 10373324.
- Blackburn AC, Tzeng HF, Anders MW, Board PG (Feb 2000). "Discovery of a functional polymorphism in human glutathione transferase zeta by expressed sequence tag database analysis". Pharmacogenetics. 10 (1): 49–57. doi:10.1097/00008571-200002000-00007. PMID 10739172.
- Polekhina G, Board PG, Blackburn AC, Parker MW (Feb 2001). "Crystal structure of maleylacetoacetate isomerase/glutathione transferase zeta reveals the molecular basis for its remarkable catalytic promiscuity". Biochemistry. 40 (6): 1567–76. doi:10.1021/bi002249z. PMID 11327815.
- Blackburn AC, Coggan M, Tzeng HF, Lantum H, Polekhina G, Parker MW, Anders MW, Board PG (Nov 2001). "GSTZ1d: a new allele of glutathione transferase zeta and maleylacetoacetate isomerase". Pharmacogenetics. 11 (8): 671–8. doi:10.1097/00008571-200111000-00005. PMID 11692075.
- Ricci G, Turella P, De Maria F, Antonini G, Nardocci L, Board PG, Parker MW, Carbonelli MG, Federici G, Caccuri AM (Aug 2004). "Binding and kinetic mechanisms of the Zeta class glutathione transferase". The Journal of Biological Chemistry. 279 (32): 33336–42. doi:10.1074/jbc.M404631200. PMID 15173170.
- Hui J, Hung LH, Heiner M, Schreiner S, Neumüller N, Reither G, Haas SA, Bindereif A (Jun 2005). "Intronic CA-repeat and CA-rich elements: a new class of regulators of mammalian alternative splicing". The EMBO Journal. 24 (11): 1988–98. doi:10.1038/sj.emboj.7600677. PMC 1142610. PMID 15889141.
- Rual JF, Venkatesan K, Hao T, Hirozane-Kishikawa T, Dricot A, Li N, Berriz GF, Gibbons FD, Dreze M, Ayivi-Guedehoussou N, Klitgord N, Simon C, Boxem M, Milstein S, Rosenberg J, Goldberg DS, Zhang LV, Wong SL, Franklin G, Li S, Albala JS, Lim J, Fraughton C, Llamosas E, Cevik S, Bex C, Lamesch P, Sikorski RS, Vandenhaute J, Zoghbi HY, Smolyar A, Bosak S, Sequerra R, Doucette-Stamm L, Cusick ME, Hill DE, Roth FP, Vidal M (Oct 2005). "Towards a proteome-scale map of the human protein-protein interaction network". Nature. 437 (7062): 1173–8. Bibcode:2005Natur.437.1173R. doi:10.1038/nature04209. PMID 16189514. S2CID 4427026.
- Fang YY, Kashkarov U, Anders MW, Board PG (May 2006). "Polymorphisms in the human glutathione transferase zeta promoter". Pharmacogenetics and Genomics. 16 (5): 307–13. doi:10.1097/01.fpc.0000205000.07054.b3. PMID 16609361. S2CID 9856864.